
Rutherford's model allowed him to make definite predictions using a simple formula he had derived about the fraction of scattered alpha particles to be found at any angle of deflection.Īrriving in Manchester in March 1912 to learn about radioactivity, it wasn't before long the 27-year-old Dane began thinking about how to prevent Rutherford's nuclear atom from collapsing. Rutherford said that such direct hits were "like trying to shoot a gnat in the Albert Hall at night". But if an alpha particle approached the nucleus head-on, the repulsive force between the two would cause it to recoil straight back like a ball bouncing off a brick wall. 
Most alpha particles would pass straight through Rutherford's atom in any "collision", since they were too far from the tiny nucleus at its heart to suffer any deflection. Rutherford's atom consisted of a tiny central core containing virtually all the atomic mass, which he later called the nucleus, but it occupied only a minute volume "like a fly in a cathedral". It led him "to devise an atom superior to J.J's" he said at time. By December 1910, Rutherford believed that given the mass and energy of an alpha particle the large deflections must be the result of a single collision with an atom. The probability that the accumulated effect of a number of tiny ricochets off electrons in Thomson's atom resulted in even one alpha particle being scattered backwards was almost zero. But Rutherford knew that the atom of his old mentor couldn't explain alpha particle scattering. Its ingredients consisted of a ball of diffuse "positive electricity" in which negatively charged electrons were embedded like plums in a pudding. The most widely-accepted atomic model was Thomson's so-called "plum pudding". In June 1909 they published their extraordinary results, but with Rutherford unable to offer any kind of explanation they attracted little interest.Īfter decades of intense arguments, by 1910 the reality of atoms was established beyond reasonable doubt. What is this proc? Single covalent bond? what would be the volume of CO2 (at STP) produced from the complete reaction of 10 grams of CaCO2? What is the formula for sulferic acid? What direction do molecules of osmosis move? Is calcium the same as limescale? Covalent bonds can best be described as a what of electronts? A 3.0-milliliter sample of hno3 solution is exactly neutralized by 6.0 milliliters of 0.50 m koh what is the molarity of the hno3 sample? What element contains 3d6? Calcium Metal Hydrochloric Acid produce? Why h2O Is liquid and H2O is gas? Why does ytterbium have the Yb symbol? With which group of elements is magnesium most reactive? How many molecules are there in 2.Marsden and Geiger made comparative measurements using different metals and they discovered exactly they same large angle scattering. This loss of electrons creates free radicals which destroy chemical bonds in the food causing it to spoil. Trending Questions Why does the melting point of cobalt high? What period is arsenic in? What type of bond is NO3? How is hydrogen found in nature? What is lead iv oxide and a sulfuric acid solution to produce an electric current the products of the reaction are lead ii sulfate in solution and water? Food can spoil through mere exposure to air because it will lose electrons. 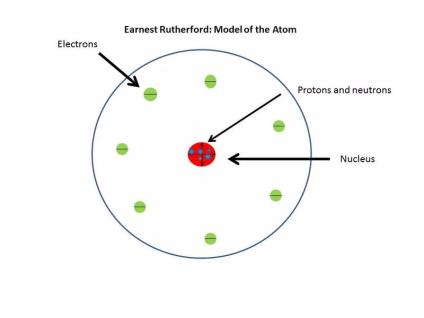
Relative size of the atomic nucleus to the distance to theĮlectrons as the 'fly in the cathedral'. Void before the electron cloud was encountered. He reasoned that the Nucleus was surrounded by a relatively large With the positively charged nucleus of the gold atom and it hadīeen rebounded due to like charges repelling. Likened this event as 'firing a 7 inch shell at a piece of tissue He then placed the detector beside the proton 'gun' and Slowly they wereīrought to the side of the foil and still his staff were detecting His detectors were atįirst place behind the foil and a large number of protons wereĭetected as flashes of light on the detector. Rutherford devised an experiment where alpha particles wereįired at a very thin sheet of gold leaf.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
